We know the No Surprises Act can be a bit confusing, but no worries, we’re here to help. In this blog post, we’ll be sharing some insider insights and provider data management tips for health plans like yours on how to meet the provider directory requirements of the No Surprises Act. Let’s dive in and discuss the right processes, protocols, and procedures together!
What Are the Provider Directory Requirements?
A process isn’t a process unless it’s documented. That’s especially true when it comes to health plan compliance. We’ve seen this play out with Medicare Advantage Triennial Reviews and now we’re seeing it with the No Surprises Act Provider Directory Requirements. The No Surprises Act requires health plans to have a documented, defendable process to show their efforts in establishing and maintaining an accurate provider directory. As a quick recap, health plans need to be able to prove that the process they have in place allows them to say:
- Yes, we have a process for provider outreach and attestation.
- Yes, we can comply with the updates to provider information.
- Yes, our process allows us to be responsive in the time required for:
- responding to the requests we receive from members about a provider’s status being in-network.
- responding to our providers who change their information.
- updating our provider directory in that 2-business day timeline
As the regulators add the finishing touches to the rule, one thing is certain: they will be looking for proof that you’re taking all the necessary steps to comply. Lucky for you, we’ve got insider tips on designing a top-notch provider data management workflow that is sure to impress the powers that be. Keep reading to get started!
Provider Data Management Tips for the No Surprises Acts
Zach Snyder, VP of Government Affairs, Quest Analytics, explains the data workflow you can implement to comply with the No Surprises Act Provider Directory Verification requirements.
Create your provider data management process
How to Set Up A Three-Tier System for Categorizing Your Providers
It’s easy to feel like there are a million and one different ways you could go about designing your provider data management process. You might be asking yourself, “Will my approach be accepted by the regulators?” Don’t worry because we’ve got your back!
Our amazing Regulatory Team has done all the legwork for you and created a data workflow that allows you to be compliant with the No Surprises Act. And the best part? It’s easy to incorporate into your existing processes.
To begin, create a three-tier system to categorize the providers in your database as the following:
- Providers who remain in the directory.
- Providers who remain in the directory, but you’ll want to flag them.
- Providers who do not stay in the directory.
Now, let’s dive into the data workflow even further to make sure you have a strong, reliable process in place. All you have to do is follow the steps and you’ll be good to go!
Tier One: Providers who remain in the directory
The No Surprises Act stipulates that health plans must have a process in place to verify and update the provider directory information at least every 90 days1 for each provider and healthcare facility included in the database. Providers and facilities who verify their information as required and the information they provide does not raise concerns, stay in the provider directory.
Tier Two: Providers who remain in the directory, but you'll want to flag them
What happens when you have providers or facilities who verify the information within the required timeframe, but some of the responses are questionable? You can add them to the directory. However, don’t stop there.
PRO TIP: We strongly suggest you flag and research the suspect responses to ensure the data you provide is reliable and accurate. Doing this will give you the peace of mind that the information you present in your directory and database is trustworthy.

Tier Three: Providers who do not stay in the directory
Now that you know when to keep providers in the directory, let’s walk through what you should do when you need to remove providers from the directory.
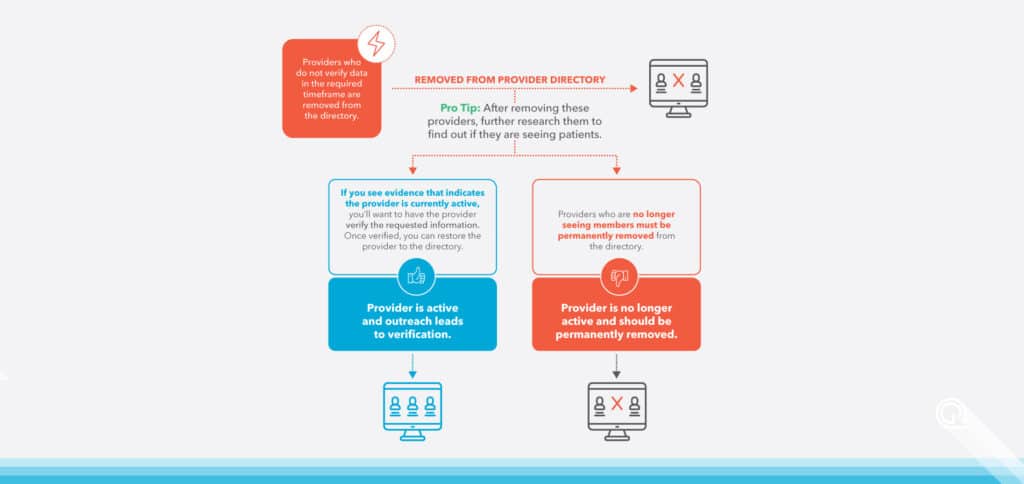
Providers must be removed from the directory when they fail to verify their data in the required timeframe. This removal is done in order to ensure that the directory remains up-to-date with the correct information regarding your network.
PRO TIP: It’s a smart move to double-check whether those providers are still seeing patients after they’re removed from the directory. Look for any hints of activity that show the provider is still active, such as claims, and then ask them to confirm the information. If they do, you can add them back to the listing.
If you determine the provider is no longer practicing, the provider must be permanently removed from the directory.
Reclaim Your Time with Quest Analytics Accuracy Services
Be Compliant and Efficient with Provider Data Accuracy
Provider data accuracy is a complex and challenging issue, which is why we provide Quest Enterprise Services® (QES®) Accuracy to handle the heavy lifting for you. From provider outreach & attestation to API capabilities and continuous compliance monitoring, we’ve got it all covered. With our actionable analytics, automated network modeling and unbeatable expertise, you can focus on achieving your ultimate objectives.
Book a free consultation today to discover how we can help you reclaim your time and achieve your provider data accuracy goals!
Footnotes
- The 90 days could change depending on the state that you operate in and the agreement between the health plan, provider and/or facility.
Proven Solutions for Your Provider Network Management
Looking to simplify your workload? Let Quest Analytics take on the heavy lifting! Our solutions and dedicated team specialize in provider data accuracy and provider network adequacy for various lines of businesses, including Medicare Advantage, Medicaid and Commercial. Schedule a strategy session today and see how we can help you every step of the way.






