Staying on top of all the latest updates in the industry can be a real challenge. That’s why we’re here to break down everything you need to know about the Qualified Health Plan (QHP) certification and network adequacy compliance. Grab your favorite beverage and get ready to dive into the world of QHP certification and network adequacy.
QHP Application and Network Adequacy Requirements
Background: Ensuring Quality and Access in Healthcare Coverage
Every year the Department of Health and Human Services (HHS) issues the Notice of Benefit and Payment Parameters (NBPP) rule, outlining the parameters for the health insurance marketplace. One of the crucial aspects covered in the rule is network adequacy standards, which health plans and stand-alone dental plans (SADPs) must meet to offer qualified plans through the Federally-facilitated Exchanges (FFEs) and State-based Exchanges on the Federal platform (SBE-FPs).
The Role of Network Adequacy for QHP Issuers
Network adequacy is about ensuring that health insurance plans provide a sufficient choice of providers and meet network adequacy year-round. In simpler terms, it’s about making sure that when people need medical or ancillary care, they can see a healthcare provider within their plan’s network.
Recognizing the significance of network adequacy in achieving healthcare equity and enhancing consumer access to affordable, high-quality care through the Exchanges, HHS stated in their Final Rule for 2023, “HHS is of the view that strong network adequacy standards are necessary to achieve greater equity in health care and enhance consumer access to quality, affordable care through the Exchanges.”
Learn more about our provider network management solutions for QHP certification.
QHP Application and Network Adequacy Reviews
CMS reviews, and approves or denies, QHP applications from issuers that are applying to offer QHPs on the FFEs1, except for FFEs in certain states.
CMS will not evaluate QHP network adequacy in FFE States performing plan management functions that elect to perform their reviews of Issuers seeking QHP certification in their state, as long as the state applies and enforces quantitative network adequacy standards that are as stringent as the Federal network adequacy standards and the network adequacy reviews are conducted before QHP certification. CMS will continue to coordinate with state authorities to address network adequacy compliance.
Network Adequacy Requirements: Provider Specialty Type, Time and Distance Standards, and County Classification
When it comes to network adequacy, the primary goal is to ensure that QHP Issuers consistently offer their members reasonable and timely access to healthcare providers. To comply with the network adequacy requirements, a QHP Issuer on an FFE must meet the following:
- Provider and Facility Specialty Types
- Time and Distance Standards
- County Classification
Want a more in-depth breakdown of the latest network adequacy requirements? Download our QHP Policy Brief.
ESSENTIAL COMMUNITY PROVIDERS
QHP Issuers must submit Essential Community Providers (ECPs) information as part of their QHP Application. Issuers must have a sufficient number and geographic distribution of ECPs, where available, in accordance with 45 CFR 156.235.
In 2022, the required ECP participation standard increased from 20 percent to 35 percent of available ECPs in the plan’s service area, including approved ECP write-ins that will also count toward a plan’s satisfaction of the 35 percent threshold.
When a plan does not meet ECP Standards, there are two options:
- Add more contracted providers to the network to come into alignment with the standards;
- Submit a completed ECP Justification Form
The justification process will require plans that do not yet meet the ECP standards to detail:
- The reasons that one or more ECP standards were not met;
- The mitigating measures the plan is taking to ensure enrollee access to ECPs; information regarding enrollee complaints regarding access to ECPs;
- And the plan’s efforts to recruit additional ECPs.
CMS will use any updated provider data and the completed ECP Justification Form submitted as part of the certification process to assess whether the plan meets the regulatory requirement, prior to making the certification decision.
For plans that use tiered networks, ECPs must be contracted within the network tier which results in the lowest cost-sharing obligation for the respective plan’s enrollees in order for those ECPs to count toward the plan’s satisfaction of each element of the ECP standard.
For plans with two network tiers, for example, participating providers and preferred providers, where cost-sharing is lower for preferred providers, only providers in the preferred network will be counted toward satisfying the ECP standards. The lowest cost tier of the network would be counted towards ECP standards.
Network Adequacy Telehealth Requirements
Issuers must report if a provider offers telehealth services within the Telehealth Services column of the Network Adequacy Provider tab. For each provider, Issuers will indicate whether that provider offers telehealth by selecting one of the following responses:
- Yes
- No
- Requested information from the provider and awaiting their response
Issuers who do not already have data on whether their providers offer telehealth will need to collect this information prior to QHP certification. QHP Issuers that do not currently collect this information may do so using the same methods they already use to collect information from their network providers relevant to time and distance standards and provider directory information. Issuers that do not have the information available at the time of the QHP certification process will be able to respond that they have requested the information from the provider and are awaiting the provider’s response.
Changes to the Network Adequacy Template
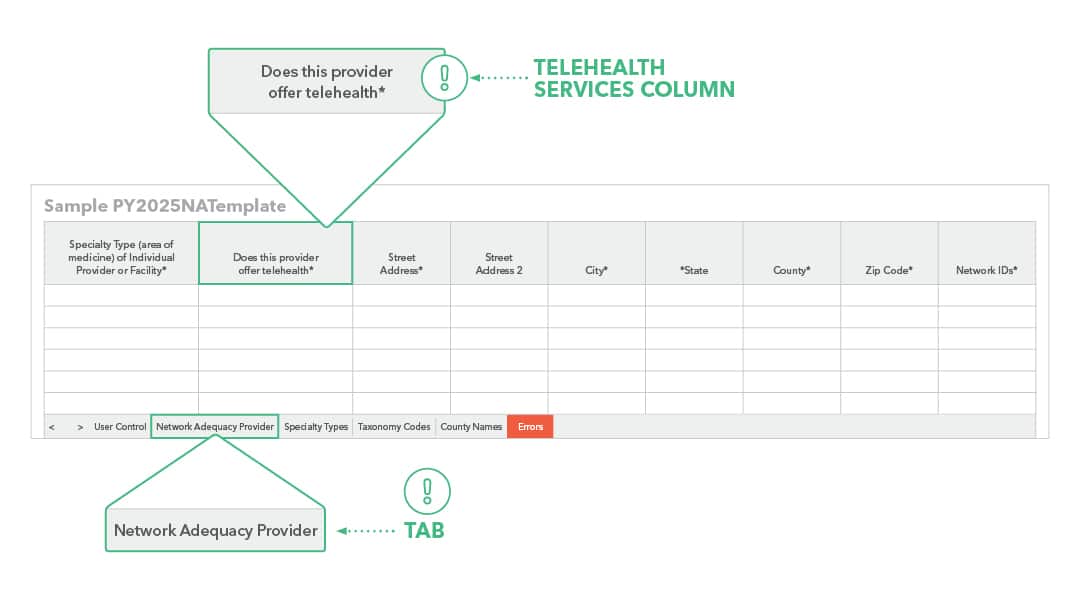
Network Adequacy Provider Tab
The Individual Providers (NA) and Facility Providers (NA) tabs from the NA Template have been combined into a new tab: Network Adequacy Provider.
Telehealth
Issuers must report if a provider offers telehealth services within the Telehealth Services column of the Network Adequacy Provider tab. For each provider, issuers will indicate whether that provider offers telehealth by selecting one of the following responses: ‘Yes,’ ‘No,’ or ‘Requested information from provider and awaiting their response.’
Download our policy brief to learn more.
APPOINTMENT WAIT TIME Standards
Beginning with the 2025 plan year (PY 2025), CMS will incorporate appointment wait time standards to assess QHP FFE network adequacy. CMS first set the Appointment Wait Time Standards in the 2023 Notice of Benefit and Payment Parameters.
The appointment wait time standards will apply to medical QHPs. For SADPs, only the dental provider specialty within the Specialty Care (Non-Urgent) category of appointment wait time standards will apply.
APPOINTMENT WAIT TIME STANDARDS FOR EXCHANGE PLAN YEAR 2025 CERTIFICATION
The appointment wait time standards measure the number of business days between when an individual requests an appointment and when the first in-person appointment is available. These standards apply to appointments for both new and existing patients.
Provider/Facility Specialty Type Appointments Must Be Available Within
- Behavioral Health 10 business days
- Primary Care (Routine) 15 business days
- Specialty Care (Non-Urgent) 30 business days
Plans would be required to meet the above standards 90 percent of the time at a minimum.
For SADPs, only the dental provider specialty within the Specialty Care (Non-Urgent) category of appointment wait time standards will apply. The dental provider specialty includes all dental providers (including general dentists and dental specialists) listed in the ECP/NA template Taxonomy Codes tab. CMS expects to issue future rules and guidance on how plans will need to demonstrate compliance with the appointment wait time standards.
A COMPLIANCE SOLUTION FOR QHP NETWORK ADEQUACY
No need to worry about manual network adequacy analysis—we’ve got you covered. We offer a template based on the current CCIIO FFE QHP Network Adequacy standards. With this template, you can:- Easily monitor the compliance of your provider networks, leaving no room for surprises.
- Identify deficiencies in your network more efficiently. We’ll help you determine what’s right, what’s wrong, and what’s missing in your provider data, offering actionable insights for corrective action.
- Streamline your provider recruitment process because, let’s face it, we can all benefit from assistance in finding the best fit for our networks.
More QHP Certification and Network Adequacy Resources
Proven Solutions for Your Provider Network Management
Looking to simplify your workload? Let Quest Analytics take on the heavy lifting! Our solutions and dedicated team specialize in provider data accuracy and provider network adequacy for various lines of businesses, including Medicare Advantage, Medicaid and Commercial. Schedule a strategy session today and see how we can help you every step of the way.













